




When chlorofluorocarbons first entered the world in the early twentieth century, they were hailed as a triumph of chemistry. Non-flammable, non-toxic, and remarkably stable, CFCs seemed like the perfect industrial compounds. They slipped into refrigerators, air conditioners, aerosol sprays, and foam production with barely a whisper of controversy. For decades, they quietly improved daily life, a technological convenience so seamless that most people never even knew these chemicals existed.
"A single kilogram of CFCs can capture and annihilate 70,000 kilograms of atmospheric ozone."
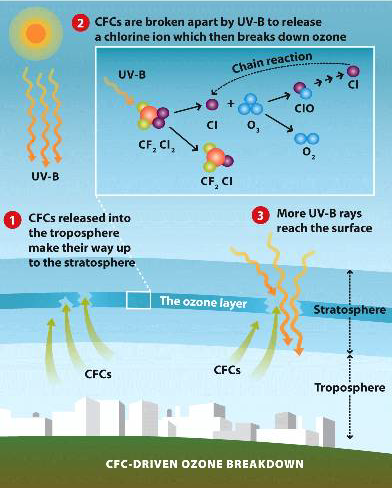
But the very qualities that made CFCs so useful - especially their chemical stability - also set the stage for an environmental crisis no one initially imagined. Once released, CFC molecules drifted upward, surviving the turbulent lower atmosphere until they reached the stratosphere. There, under intense ultraviolet radiation, they broke apart and released chlorine atoms. Each chlorine atom became a tiny saboteur, capable of destroying tens of thousands of ozone molecules through a chain reaction that unfolded invisibly above the planet.
By the 1980s, scientists had pieced together the consequences: the protective ozone layer, Earth's natural sunscreen, was thinning. The discovery of the Antarctic ozone hole shocked the world. Suddenly, the abstract chemistry of CFCs translated into tangible risks - higher UV exposure, increased skin cancer rates, weakened immune systems, and threats to ecosystems from phytoplankton to crops. What had once been a symbol of technological progress became a cautionary tale about unintended consequences.
Yet the story of CFCs is also one of rare global cooperation. When the science became undeniable, nations acted. The 1987 Montreal Protocol, often celebrated as the most successful environmental treaty in history, committed countries to phasing out CFCs and related ozone-depleting substances. Industry adapted, alternatives were developed, and the ozone layer began a slow but measurable recovery. It remains one of the clearest examples of how evidence-based policy, international collaboration, and public pressure can converge to address a planetary threat.
Today, the legacy of CFCs continues to shape environmental policy. Their rise and fall remind us that technological innovation carries responsibilities, that atmospheric chemistry is not distant from human health, and that global problems demand global solutions. The ozone layer's healing is far from complete, but its trajectory offers something rare in environmental narratives: a sense of hope grounded in collective action.