




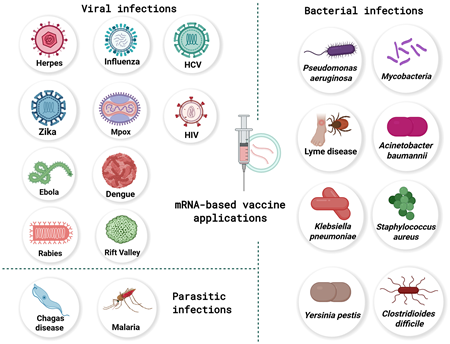
Messenger RNA (mRNA) vaccine technology, once an experimental platform, has rapidly evolved into one of the most dynamic frontiers in modern medicine. As we move through 2025 and into early 2026, researchers and public health leaders describe the field as entering a "remarkable transformation", driven by scientific breakthroughs, expanded clinical pipelines, and new applications far beyond the COVID-19 pandemic.
"The delivery of the first made-in-Canada mRNA vaccines to provinces and territories marks a major milestone in our partnership with Canada."
The success of the first COVID-19 mRNA vaccines catalyzed unprecedented global investment, accelerating the maturation of the platform. Today, roughly 70% of active mRNA vaccine trials target diseases other than COVID-19, including cancer, HIV, influenza, and a range of emerging infectious threats. This shift reflects both scientific confidence and the adaptability of mRNA constructs, which can be rapidly redesigned in response to viral evolution or newly identified pathogens.
One of the most notable recent developments is the expansion of mRNA vaccines into seasonal respiratory diseases. Moderna's new flu vaccine candidate, mRNA-100, has advanced after the U.S. Food and Drug Administration reversed an earlier decision and agreed to review the application. A 40,000-person clinical trial showed the vaccine outperformed a standard flu shot in adults over 50, signaling that mRNA platforms may soon reshape annual influenza prevention.
At the same time, researchers continue refining the underlying technology. Advances in lipid nanoparticle (LNP) delivery, codon optimization, and nucleoside modification have improved stability, translation efficiency, and immune response tailoring. These innovations are helping overcome early limitations such as reactogenicity, storage challenges, and variability in immune responses among older adults or immunocompromised individuals.
Beyond infectious diseases, mRNA vaccines are making strides in therapeutic areas once considered out of reach. Early-phase HIV mRNA vaccines have shown promising immune activation, and personalized cancer vaccines - designed to target tumor-specific antigens - are progressing through clinical trials. These efforts build on decades of foundational research that predated COVID-19 but have now accelerated thanks to global recognition of mRNA's potential.
Regulatory agencies and scientific institutions are also adapting. The FDA continues to refine its framework for evaluating mRNA vaccine durability, safety, and strain-update processes, recognizing that the platform's speed and flexibility require equally agile oversight.
Taken together, the most recent developments illustrate a field in full bloom. mRNA vaccines are no longer defined by their pandemic origins; instead, they are emerging as a versatile, rapidly deployable, and increasingly sophisticated toolset poised to transform global health. From seasonal flu to HIV, from cancer immunotherapy to pandemic preparedness, the next generation of mRNA vaccines is expanding the boundaries of what preventive and therapeutic medicine can achieve.
| • | An mRNA Crime |
| • | Mapping global public perspectives on mRNA vaccines and therapeutics |
| • | Can mRNA Vaccines Help Treat Cancer? |
| • | Understanding mRNA vaccine technologies |